
Upon store loading, a number of decisions regarding the tubers are taken. Wet tubers are dried, warm tubers are cooled and cold tubers are warmed. If tubers of different temperatures are taken in, internal air is circulated until all tubers have the same temperature, subsequently the storage compartment is brought at a lower temperature.
Where there is risk of reducing sugar accumulation, the store keepers take their time to cool. Wound healing is quickest at a high relative humidity and at 16–18 °C, not warmer nor cooler. But to avoid silver scurf (Helminthosporium solani) a quick drying is required. Quick drying is also essential when the crop contains rotten tubers to avoid them contaminating neighboring tubers through leaking.
Conditioning the tubers
The consequences of all of this are taken into consideration when loading the store and conditioning the tubers for the rest of the storage period. Store keepers keep in mind that when drying tubers, water evaporates which also cools the tubers. Evaporation is an energy costing process and the energy is delivered by the tubers. Inversely, if the store keeper intends to cool the tubers, they are dried.
Relative humidity
The Mollier diagram is used to find out what kind of air (temperature and relative humidity (RH) wise) has drying capability. Say the outside temperature is 15 °C and has a RH of 60%, then it contains 7.6 g of water per m3. With an inside temperature of 11 °C and a RH of 90% (inserted, assuming that measuring RH values over 90% is unreliable), the air contains 8.9 g water per m3, which is more than the outside air. So, introduced outside air is able to absorb more water and will dry the inside air, possibly adhering water. If only cooling is desired and not drying, a moisture content with a number of g/m3 with little difference is chosen earlier or later in the day or upon refrigeration.
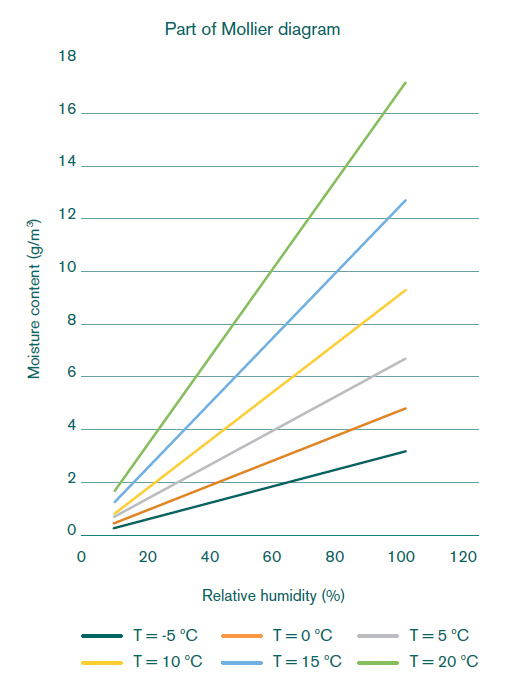
German physicist Richard Mollier (1863–1936) developed enthalpy-entropy charts which are used to calculate the maximum amount of water vapor that air contains at each temperature. If the temperature drops, water vapor condensates to form liquid water; if the temperature rises, it has a drying capacity. To manage potato store humidity and temperature, these relations are of invaluable interest.
Want to learn everything about the potato? Our Potato handbook is a global potato university in 1 book!
Buy your copy today and get a 1 year free subscription to Potato world magazine.

